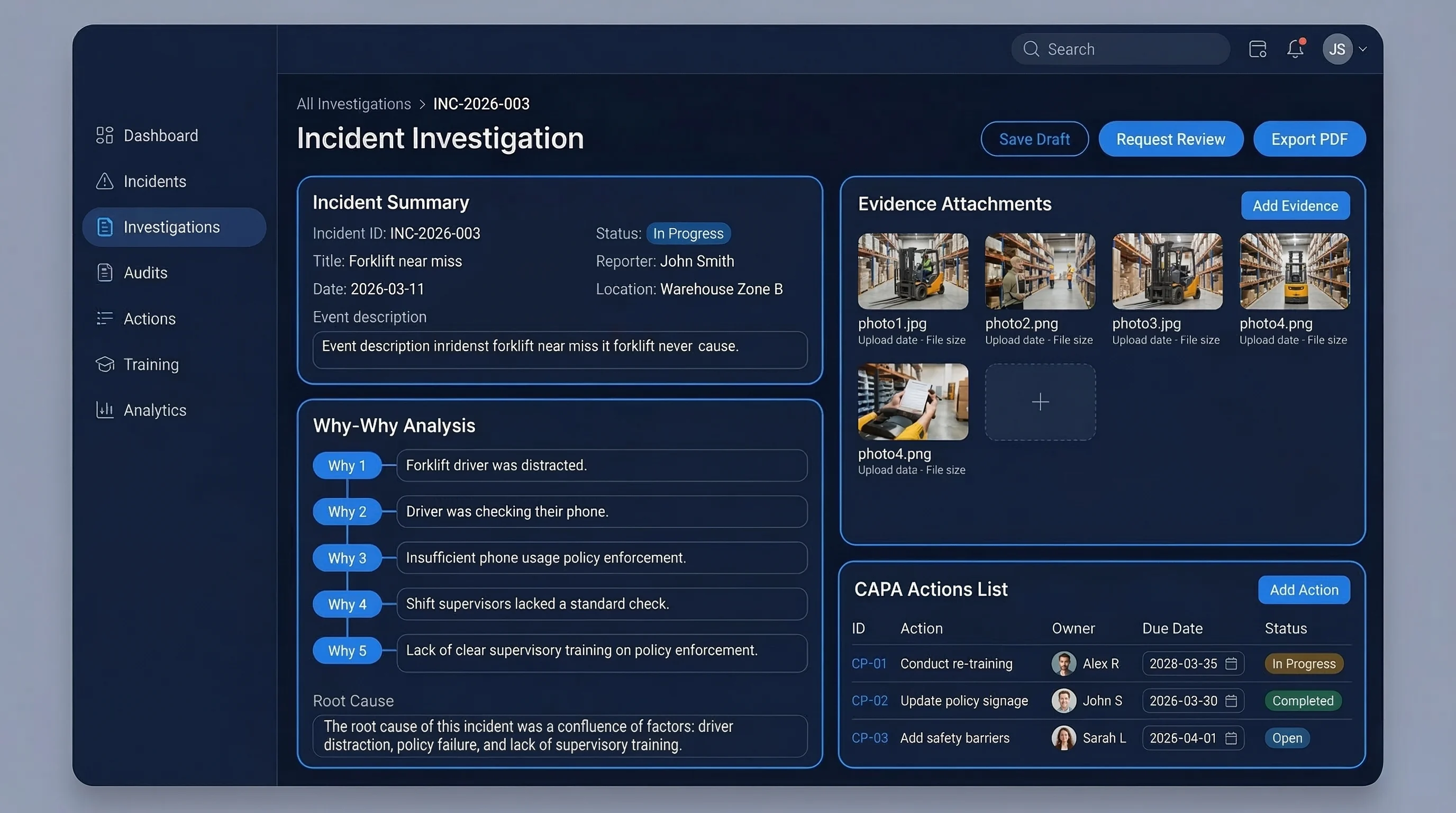
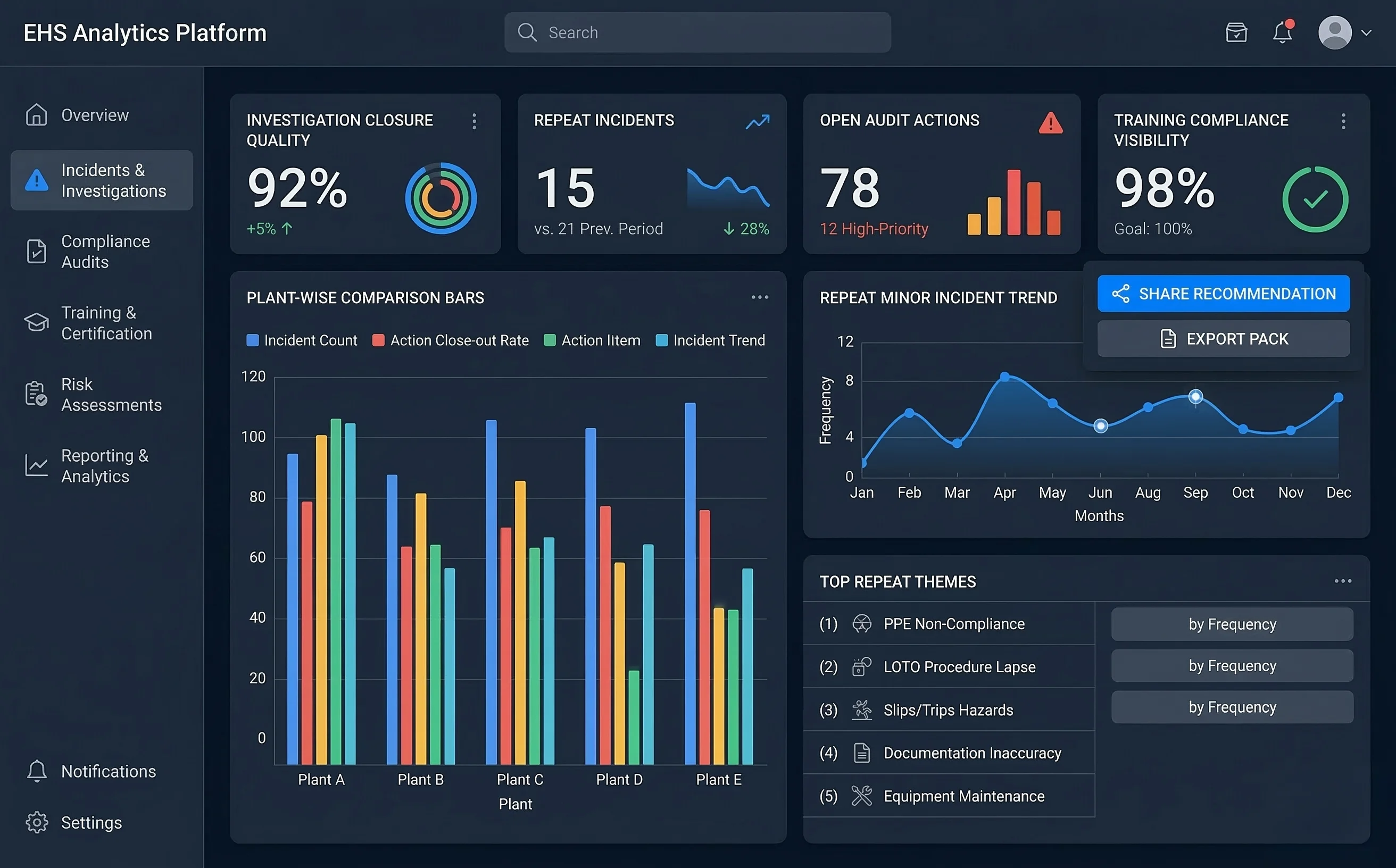
Investigation closures became predictable across automated food plants
A large food processing & FMCG manufacturer replaced ad-hoc investigations, manual audit tracking, and fragmented training records with structured RCA workflows, action automation, and cross-plant dashboards.